When diving, everyone experiences a vague fear of deep water at least once. The dark blue at 30 or 40 meters down is enough to impose psychological pressure. Conversely, shallow waters around 5 meters provide us with a great sense of relief, thinking we can pop our heads out of the water at any time.
However, from the perspective of physics, this intuition is completely wrong. Setting aside physiological risks like nitrogen narcosis or decompression sickness, and solely looking at the risk of "gas expansion" causing direct physical damage to a diver's lungs, the most dangerous zone in the ocean is not the deep water, but the 10-meter zone right below the surface. Through Boyle's Law, we will uncover the terrifying paradox hidden in shallow waters.
The Seesaw Game of Pressure and Volume: Boyle's Law
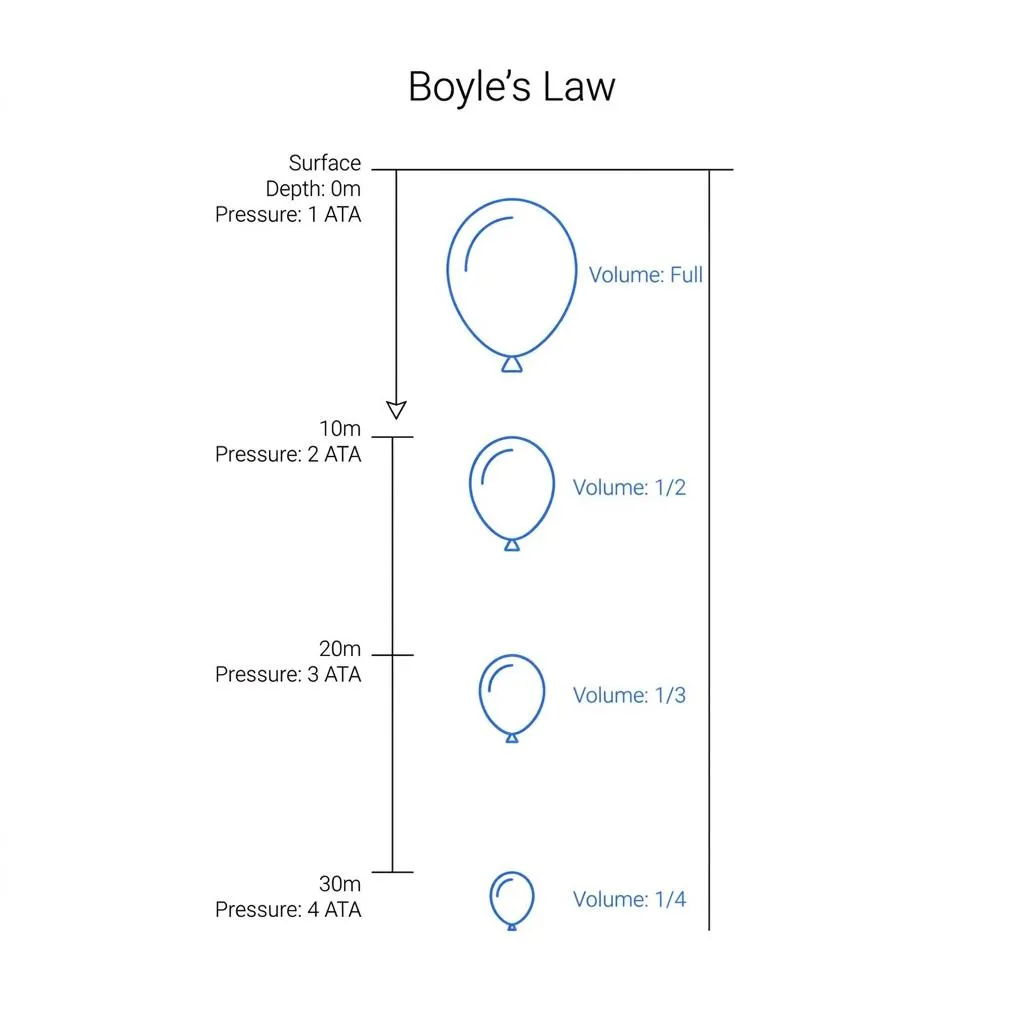
The most powerful and simple principle governing the underwater world is Boyle's Law. This law, stating that the volume (V) of a gas is inversely proportional to pressure (P) when temperature is constant, is expressed by the following simple equation:
The surface pressure where we live our daily lives is 1 atmosphere (1 ATA). Since water pressure increases by 1 ATA for every 10 meters we descend, the pressure is 2 ATA at 10 meters, 3 ATA at 20 meters, and 4 ATA at 30 meters. As the pressure doubles, triples, or quadruples, the volume of air inside our lungs or BCD inversely shrinks to 1/2, 1/3, and 1/4. Up to this point, this is basic knowledge everyone learns in Open Water training.
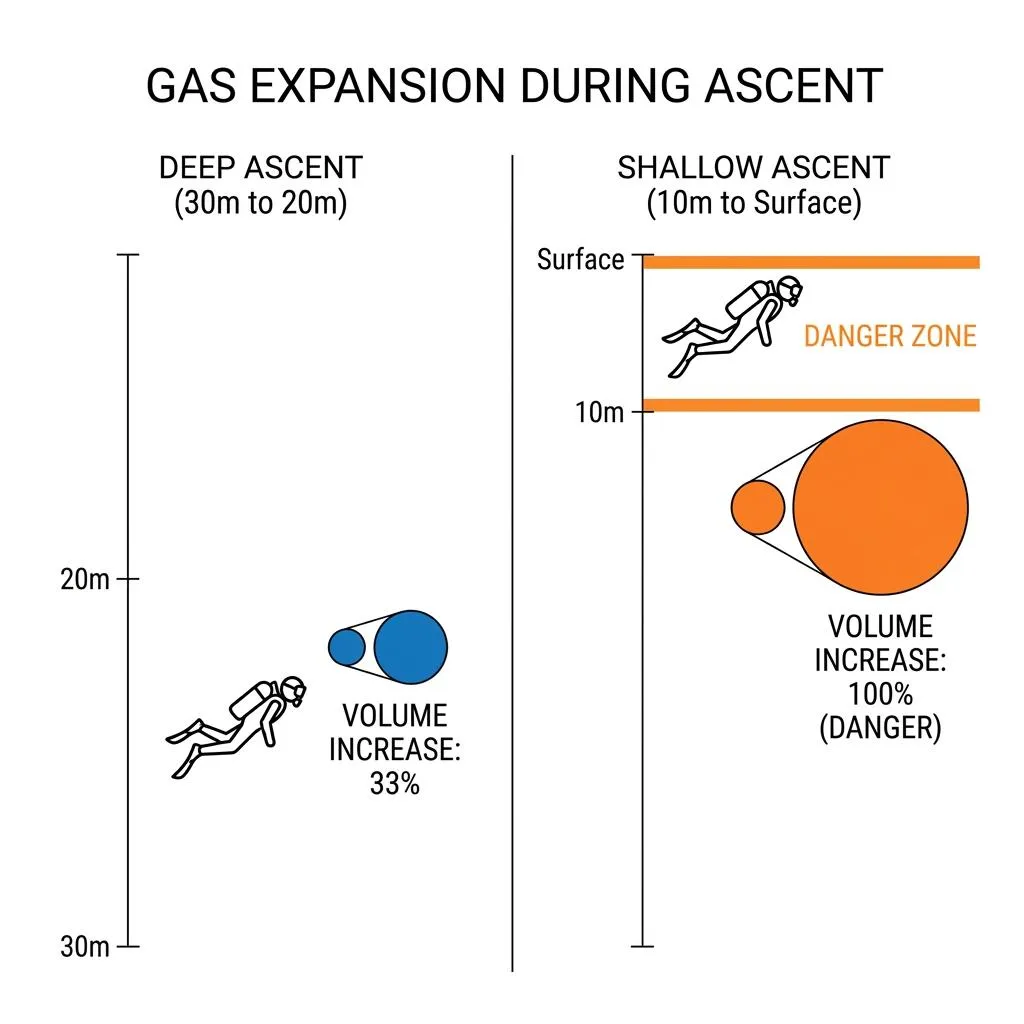
Ascending in the Deep vs. Ascending in Shallow Water
The real issue occurs when we "ascend" to the surface. Suppose we ascend 10 meters from a depth of 30 meters (4 ATA) to 20 meters (3 ATA). The air volume in the lungs expands from 1/4 to 1/3. Numerically, the volume expands by about 33%. This is a gentle expansion that we can easily handle and exhale through normal breathing.
This time, let's ascend the exact same 10 meters, but from a depth of 10 meters (2 ATA) to the surface (1 ATA). At this point, the air volume in the lungs expands from 1/2 to 1. This is a whopping 100% expansion—the volume of air exactly and explosively doubles. Even though the distance traveled is the same 10 meters, the air that expanded by 33% in the deep water expands by 100% in shallow water. This is precisely the paradox of the deep water.
Our Mindset Toward the 5-Meter Depth, the Danger Zone
What happens if a beginner diver, after finishing their dive and doing a safety stop at 5 meters, panics due to low air supply or a failure in buoyancy control, holds their breath, and shoots up to the surface? With an ascent of just 5 meters, the air in the lungs expands rapidly by nearly 1.5 times. If the ballooning air cannot find a way out, it will eventually tear the alveoli and enter the bloodstream, causing a fatal Arterial Gas Embolism (AGE).
This is the exact physical reason why the shallow water just below the surface, which seems the safest, is cited as the most lethal zone in diving accident statistics.
You should now understand why we must strictly control our ascent rate to 9 meters per minute or less, and why the 3-minute safety stop at 5 meters is not simply for off-gassing nitrogen from the body, but an essential buffering process to adapt the body to explosive pressure changes. On your next dive, rather than relaxing when you reach shallow depths, recognize that it is the space where the most dramatic physical changes are occurring, and try to exhale long and smoothly until your very last breath.